Case Study
Case Study
Context
Just as the development and use of monoclonal antibodies has revolutionized treatment of cancer, monoclonal antibodies are increasingly recognized as potential therapeutics for the treatment of infectious disease. However, as with vaccines, the lack of detailed knowledge of the correlates of protection has hindered the development of effective monoclonal therapeutics for the treatment of infectious disease: of the almost 80 approved monoclonal antibody therapeutics, only 4 have been approved for infectious disease. Importantly, however, just as the correlates of protection identified using Systems Serology can be used to guide vaccine design, Systems Serology can be used to identify the mechanism of action of monoclonal therapeutics, which can then be used to develop more effective therapeutics.
Problem
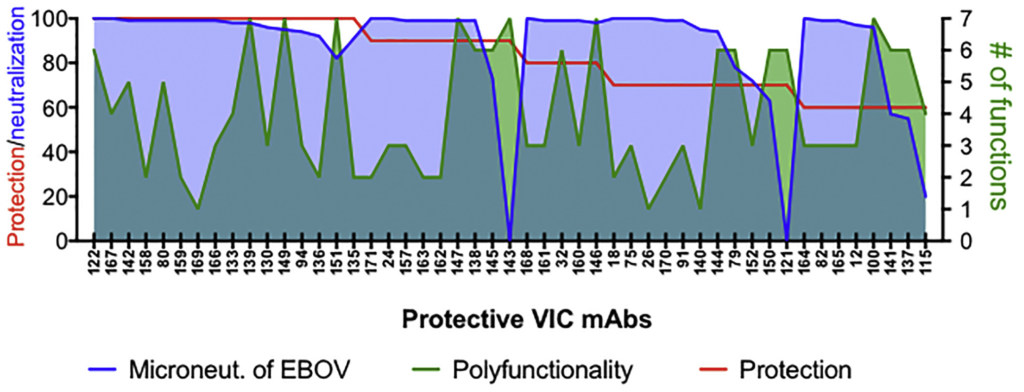
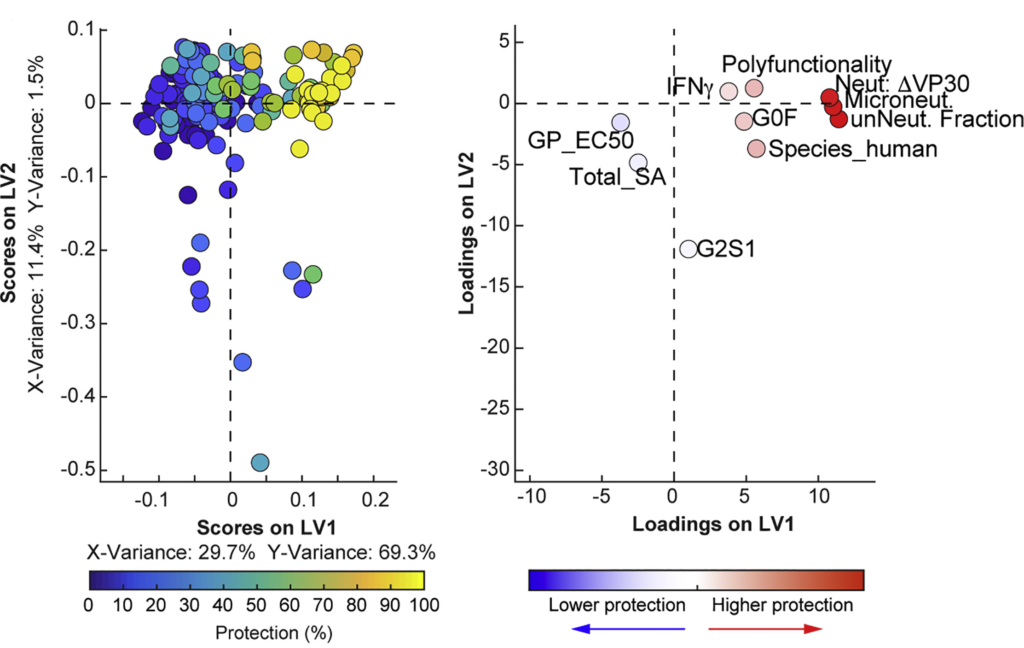
For many infectious disease targets, neutralization is often the primary means by which antibody candidates are selected for clinical development. However, as with the mounting importance of extra-neutralizing antibody functions in vaccine-mediated protection from infection, extra-neutralizing antibody functions are increasingly recognized as critical in monoclonal antibody–mediated protection from infection. In numerous studies with both individual antibodies and cocktails of antibodies, non-neutralizing antibodies have demonstrated protection in animal models of Ebola virus infection. Coupled with the observation that not all neutralizing antibodies are protective, this suggests that neutralization activity is neither necessary nor sufficient for protection in vivo. However, defining the functions of EBOV-specific monoclonal antibodies that are linked to protection had proven difficult. As part of the Viral Hemorrhagic Fever Immunotherapeutic Consortium, Systems Serology was used to profile almost 170 Ebola-specific monoclonal antibodies to define the antibody features and functions critical to protection.
Systems Serology Application
While neutralizing activity was clearly linked with protection, analysis of antibody effector functions suggested a role for additional Ab functions in protection. Critically, for monoclonal antibodies with limited neutralizing activity, increased extra-neutralizing antibody functions were associated with protection in vivo. In particular, the ability of antibodies to induce phagocytosis and activate NK cells were particularly important for protection by non-neutralizing antibodies. Furthermore, neutralizing antibodies that lacked the ability to induce phagocytosis were less protective in vivo, suggesting that neutralization may not be the critical functional activity underlying protection in vivo. Similar observations have been observed in HIV and influenza infection, further highlighting the importance of extra-neutralizing antibody functions in monoclonal antibody–mediated protection. Ultimately, the data generated from this study provides a critical framework to both define and exploit immune correlates of protection to rationally guide the development of effective therapeutics against Ebola virus and other human pathogens.
Conclusion
The comprehensive profiling of Ebola virus–specific monoclonal antibodies using Systems Serology ultimately identified functions key to protection. Critically, the identification of these functions provides the roadmap to increasing the efficacy of existing therapeutics through enhancement of functional activity. Furthermore, the incorporation of assays probing these functional correlates into the screening process used to down select therapeutic candidates, could accelerate the development of rationally designed and functionally enhanced monoclonal antibody–based therapies. Given the important role monoclonal therapeutics will play in slowing emerging infectious epidemics, the Systems Serology suite of assays offered by SeromYx provides a platform in which antibody functionality can be broadly and rapidly determined, ultimately providing critical information for the selection of highly functional monoclonal antibodies that should move further into clinical development.
Reference
Bronwyn M. Gunn, et al., A Role for Fc Function in Therapeutic Monoclonal Antibody-Mediated Protection against Ebola Virus. Cell Host Microbe. 2018 Aug 8; 24(2): 221–233.e5.